Gyros Protein Technologies (formerly Gyros AB) is in utilization of microfluidic technologies to miniaturize and automate immunoassays. Gyrolab immunoassay platforms increase productivity and efficiency during development of biotherapeutics and vaccines, saving time, labor and sample. Major biopharmaceutical companies and their service providers utilize Gyrolab platforms to boost time-critical workflows and meet increasingly stringent regulatory demands.
Whether you need to accelerate assay development for preclinical R&D, facilitate upstream and downstream bioprocessing or meet critical data and time requirements for regulated bioanalysis, Gyros will put a spin on what’s possible in your laboratory. Using nanoliter microfluidics, Gyros’ flow-through technology simplifies assay workflows by eliminating incubations and shortening run times to produce reliable data exhibiting 3- to 4-log dynamic range and minimal matrix interference.
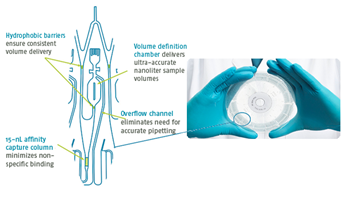
Fig 1. CD with technology of nanoliter microfluidics
Features:
Time Saving: Analyze one CD in one hour, develop new analytical method in several days rather in several weeks.
Sample Volume: Utilize technology of nanoliter microfluidics, one mouse for one PK curve, reduce animal usage in preclinical studies.
Throughput: Generate up to 96/112 high quality data points per CD, run five CD in one batch.
Detection Range: Lower LLOQ and higher ULOQ, suitable for samples of different concentration.
Matrix Interference: Suitable for almost any samples, reduce MRD to 1/2 of original data.
Automation: Run automatically for loading samples, loading reagents, washing and detecting, avoid the personal error, reduce CV, can run unattendedly overnight and increase the throughput.
Sensitivity: pg/mL level sensitivity, suitable for sample of low concentration.
Precision and Good Repetitiveness: The results is consistent between different researchers, laboratories and companies. Increase the success rate and reduce number of replication.
Applications:
(1) Pharmacokinetics (PK); Toxicokinetics (TK);
(2) Pharmacodynamics (PD); Biomarker Monitoring;
(3) Immunogenicity; ADA Study;
(4) Product Qualification (IgG Titer) in Bioprocessing;
(5) Impurity Testing (HCP, Protein A) in Bioprocessing;
(6) Affinity Detection and Vaccine Research.
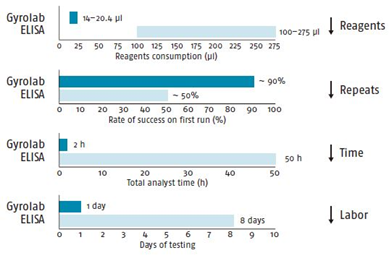
Fig. 2 Comparison of Gyrolab vs. ELISA
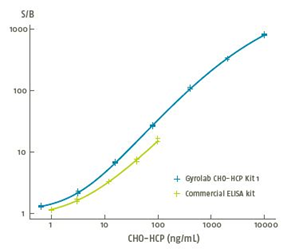
Fig. 3 Comparison of dynamic range of Gyrolab vs. ELISA
Evaluator, the software of Gyrolab xPand workstation, supports automatic curve fitting, parameters and results output and support results output to LIMS system. This software is also designed for 21 CFR part 11 compliance.
These features and advantages liberate the experimenter's hands, allowing the experimenter to develop the method with the least amount of samples in the shortest possible time, and get the best data from experiments such as methods validation and sample testing. This allows fast and easy access to data that can be used for decision-making, thereby advancing drug discovery to the next step and listing as soon as possible.
These advantages make Gyrolab platform become the major large molecule drug research platform for pharmaceutical companies and CROs, including Pfizer, Novartis, Amgen, AstraZeneca, Roche, Sanofi, Eli Lilly, etc., will become equipments for large molecule drug development in the future.
(Please refer to the product mannual for information details.)





